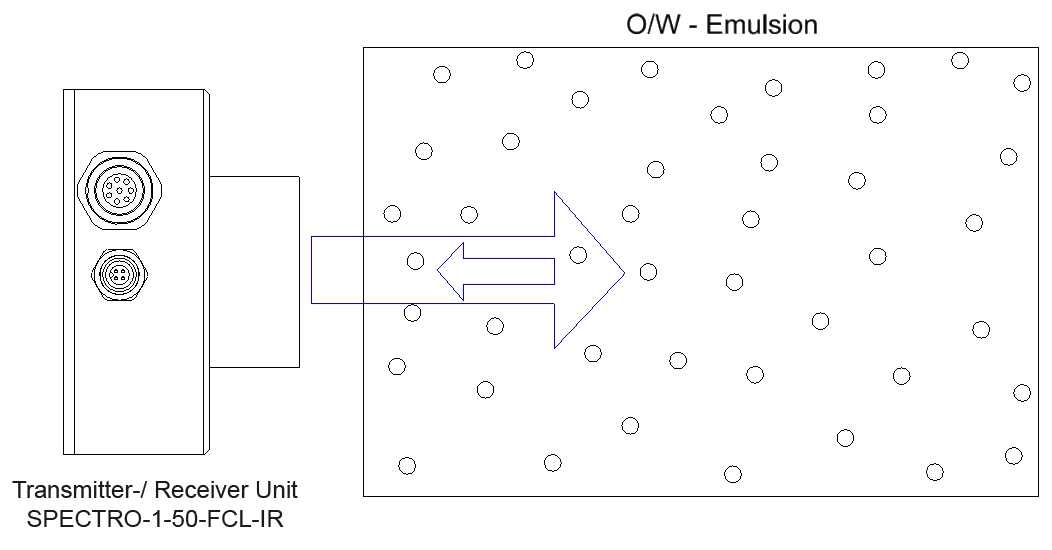
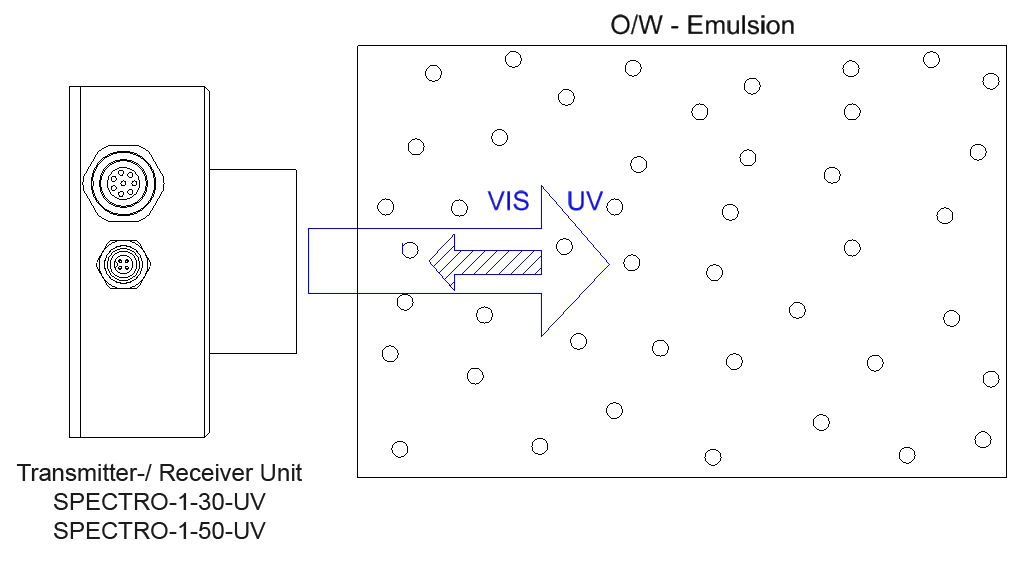
What is an emulsion?
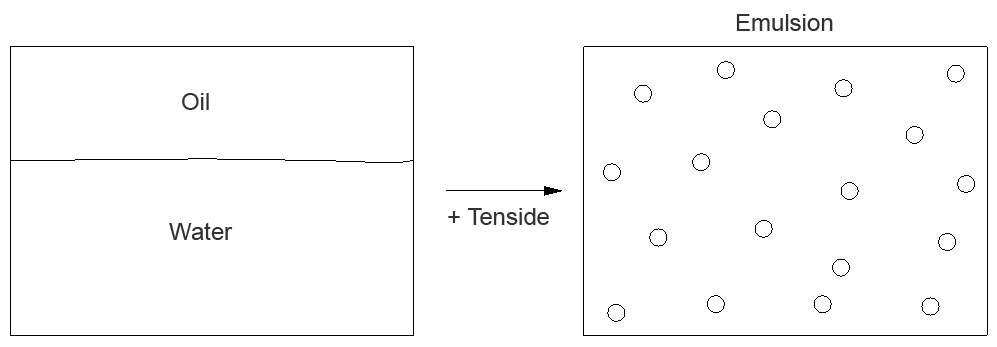
An emulsion is a homogeneous mixture of two usually immiscible liquids (e.g. oil and water). It exists in the form of a finely dispersed mixture of two liquids, where one liquid (e.g. oil) forms small droplets (dispersed or inner phase) and the other liquid (e.g. water) takes up various droplets and causes them to float (continuous or outer phase). The existence of droplets also explains the turbid, milky appearance of emulsions. In case of emulsions of water and oil both the oil (oil-in-water emulsion, O/W emulsion) and the water (water-in-oil emulsion, W/O emulsion) can form the inner phase.
Since the hydrophilic character of water and the lipophilic behaviour of oil do not show any tendency to mix, emulsifiers (so-called surface-active agents) are used to prepare and stabilise an emulsion.
Determining the phase-volume ratio
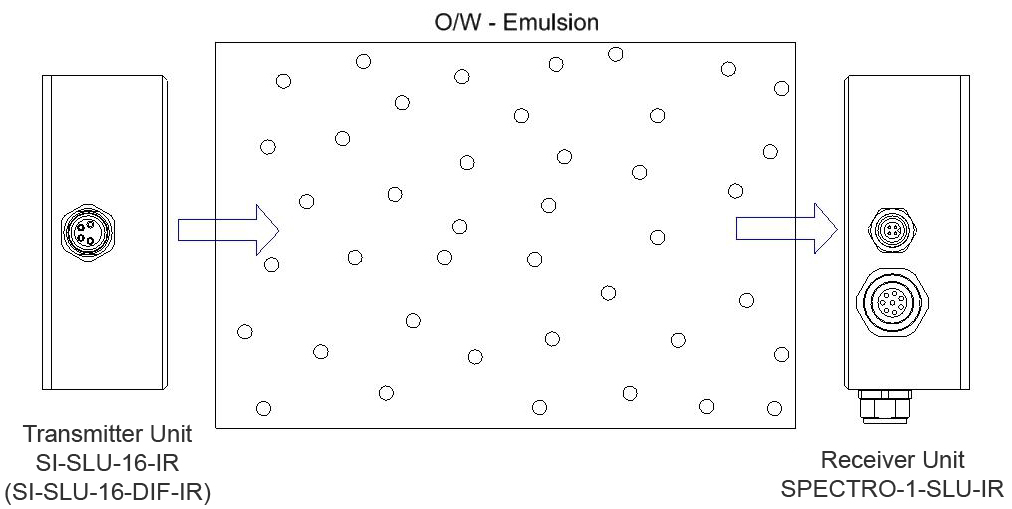
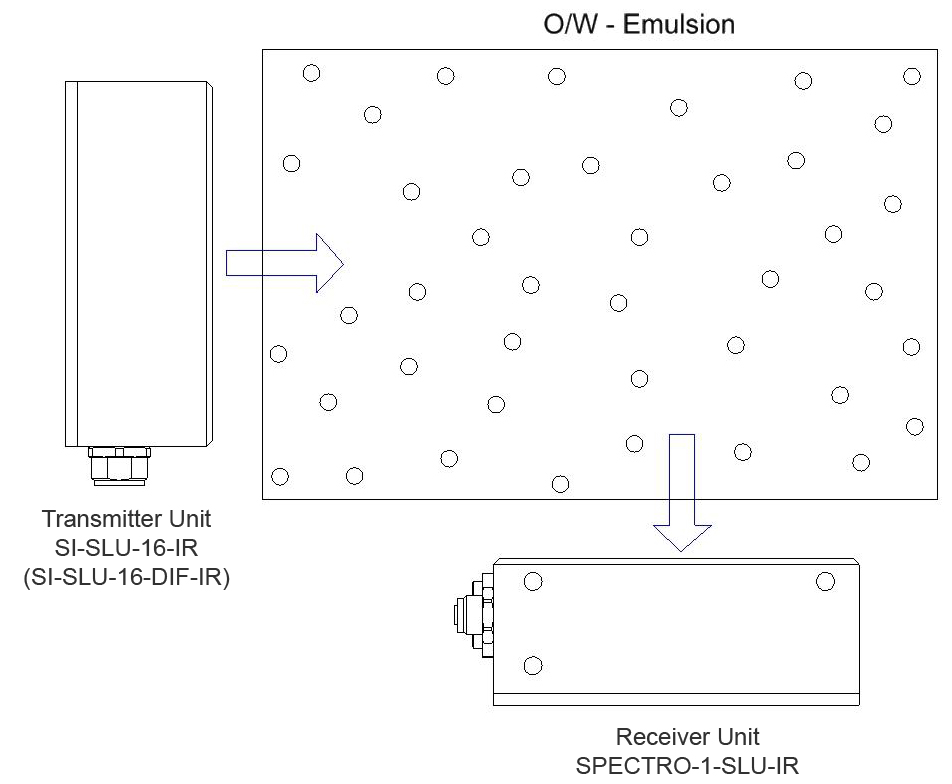
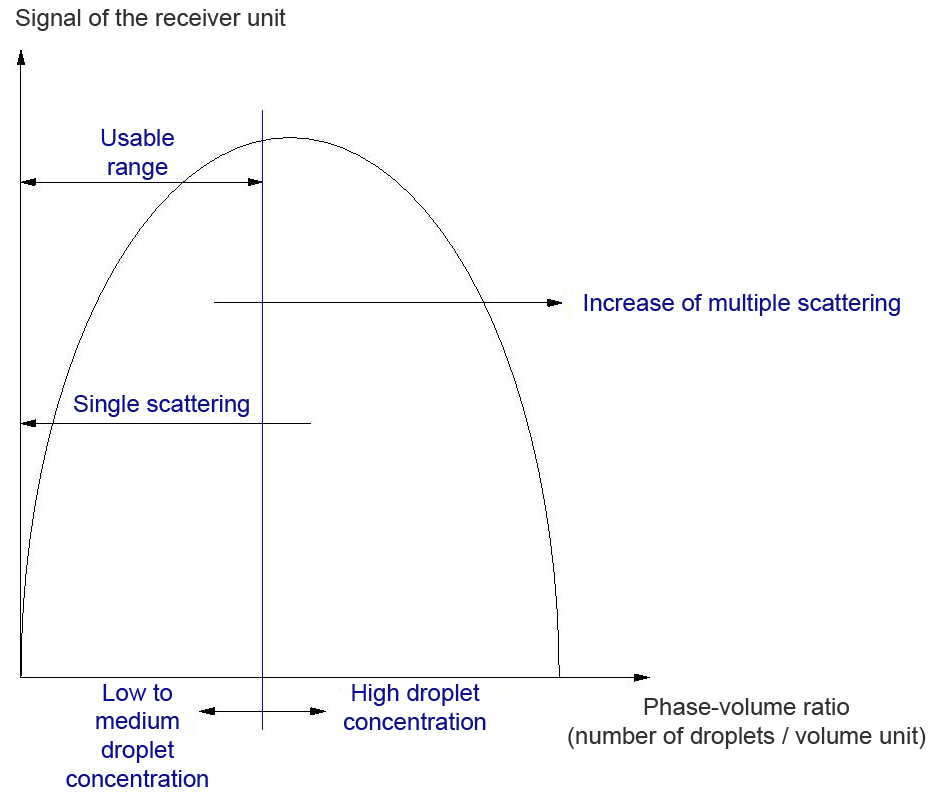
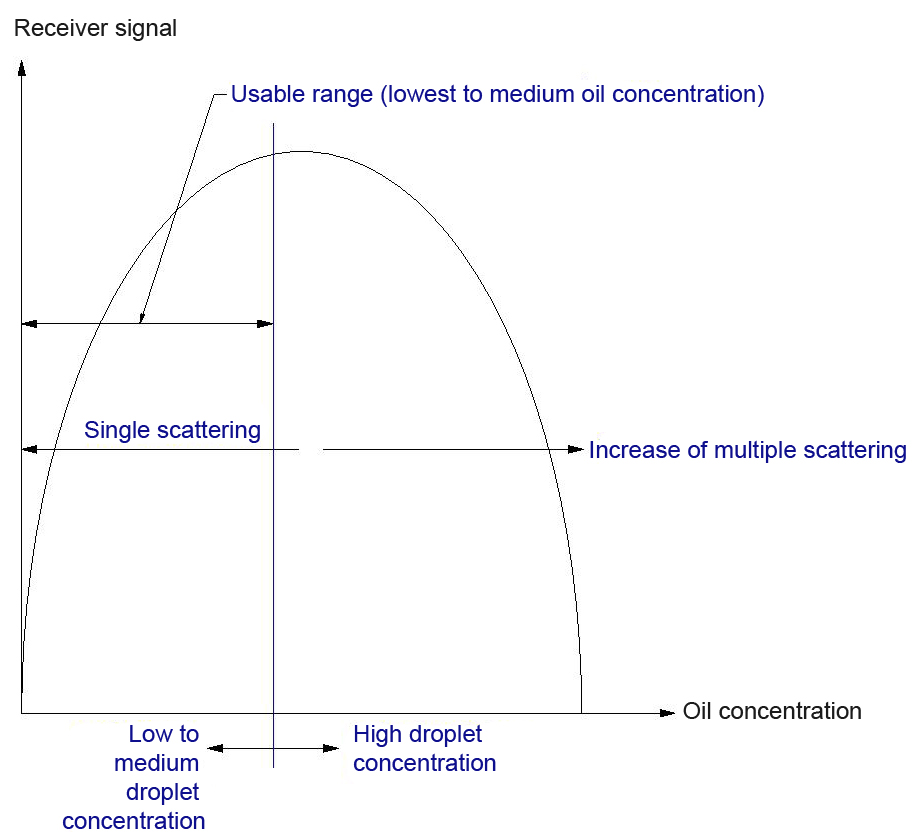
The phase-volume ratio is the quotient of the volume fraction of the inner phase and the volume fraction of the outer phase. Based on the assumption that the average particle diameter of the droplets of the inner phase during a change of the phase-volume ratio corresponds with, turbidity measurement can provide information about the phase-volume ratio.
| SPECTRO-1-SLU-IR | .jpg) |
| SPECTRO-1-50-FCL-IR | .jpg) |
| SI-SLU-16-IR | .jpg) |
| SI-SLU-16-DIF-IR | .jpg) |
Determining the oil fraction of an O/W or W/O emulsion
Emulsions on mineral-oil or vegetable-oil basis can be excited to fluoresce by using UV light.